LAPAROSCOPIC
TOTAL AND PARTIAL NEPHRECTOMY
(
Download pdf )
BENJAMIN R. LEE
Department of Urology, Long Island Jewish Medical Center, New Hyde Park, New York, USA
ABSTRACT
Laparoscopic
radical nephrectomy has established its role as a standard of care for
the management of renal neoplasms. Long term follow-up has demonstrated
laparoscopic radical nephrectomy has shorter patient hospitalization and
effective cancer control, with no significant difference in survival compared
with open radical nephrectomy. For renal masses less than 4cm, partial
nephrectomy is indicated for patients with a solitary kidney or who demonstrate
impairment of contralateral renal function. The major technical issue
for success of laparoscopic partial nephrectomy is bleeding control and
several techniques have been developed to achieve better hemostatic control.
Development of new laparoscopic techniques
for partial nephrectomy can be divided into 2 categories: hilar control
and warm ischemia vs. no hilar control. Development of a laparoscopic
Satinsky clamp has achieved en bloc control of the renal hilum in order
to allow cold knife excision of the mass, with laparoscopic repair of
the collecting system, if needed. Combination of laparoscopic partial
nephrectomy with ablative techniques has achieved successful excision
of renal masses with adequate hemostasis without hilar clamping. Other
techniques without hilar control have been investigated and included the
use of a microwave tissue coagulator.
In conclusion, laparoscopic radical nephrectomy
for renal cell carcinoma has clearly demonstrated low morbidity and equivalent
cancer control. The rates for local recurrences and metastatic spread
are low and actuarial survival high. Furthermore, laparoscopic partial
nephrectomy has demonstrated to be technically feasible, with low morbidity.
With short term outcomes demonstrating laparoscopic partial nephrectomy
as an efficacious procedure, the role of laparoscopic partial nephrectomy
should continue to increase.
Key words:
kidney; nephrectomy; laparoscopy; hemostatic techniques
Int Braz J Urol. 2002; 28: 504-9
INTRODUCTION
Laparoscopic radical nephrectomy has established its role as a standard of care for the management of renal neoplasms. Long term follow-up has demonstrated laparoscopic radical nephrectomy has shorter patient hospitalization and effective cancer control with no significant difference in survival compared with open radical nephrectomy (1). For renal masses less than 4cm, partial nephrectomy is indicated for patients with a solitary kidney or who demonstrate impairment of contralateral renal function. Open partial nephrectomy has an overall local recurrence rate of 0-10% (2). The major technical issue for success of laparoscopic partial nephrectomy is bleeding control. Several techniques including radiofrequency pretreatment, laparoscopic hilar clamping with bulldog clamps or Satinsky clamp, argon beam coagulation, electrocautery, harmonic scalpel, fibrin glue, ultrasonic dissection, Surgicel™, Avitene™, fibrin-soaked Gelfoam™ activated by thrombin, pledget reinforced sutures, hydrojet dissection, microwave tissue coagulation, and cable ties have been developed to achieve better hemostatic control. Development of new laparoscopic techniques for partial nephrectomy can be divided into 2 categories: hilar control and warm ischemia vs. no hilar control.
TECHNIQUE
Preoperative
workup includes abdominal computed tomography (CT) scan with intravenous
contrast, in order to delineate anatomy. Staging workup further includes
chest X-ray, electrolyte panel, CBC, and liver function tests. If the
alkaline phosphatase is increased, a bone scan is necessary to assess
metastatic disease. A renal scan determines percent function. If renal
function is less than 10 percent, the patient is better served with radical
nephrectomy.
A 5F ureteral catheter is cystoscopically
placed at the beginning of the case, to allow retrograde injection after
excision of the mass to determine if the collecting system has been violated.
The ureteral catheter is tied to a 16F Foley catheter with a silk tie.
A 60cc syringe with dilute indigo carmine is affixed to the ureteral catheter
for subsequent retrograde injection.
Three trocars are used in a transperitoneal
approach. After initial insufflation with a Veress needle, an 11mm trocar
is placed under direct visualization using the Optiview trocar (Ethicon,
Cincinati, Ohio). This trocar has a cutting element, which dissects through
fascia under direct visualization to perform the pneumoperitoneum. The
second 12mm trocar is placed lateral to the rectus in the midclavicular
line, at the level of the umbilicus. The third 5mm trocar is placed halfway
between xyphoid process and umbilicus. After incising along the line of
Toldt, the colon is reflected medially. On the right, the lateral colonic
peritoneal reflection is incised from the right common iliac artery up
to the hepatic flexure. The anterolateral surface of the right kidney
is often not entirely behind the ascending colon, and is usually covered
by the lateral peritoneum. The right triangular and anterior coronary
ligaments must be divided. The colorenal attachments must then be sharply
divided to allow the ascending colon and hepatic flexure to be rolled
medially. The duodenum is exposed and then mobilized medially, by means
of the Kocher maneuver, until the vena cava is clearly visualized. On
the left, mobilization must take place from the splenic flexure down to
the level of the common iliacs.
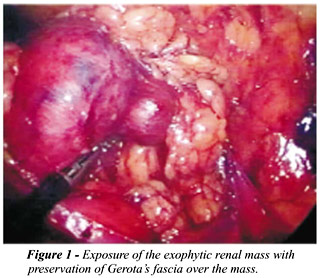
Exophytic renal masses can often be localized
by mobilizing the kidney within Gerota’s fascia, being conscious
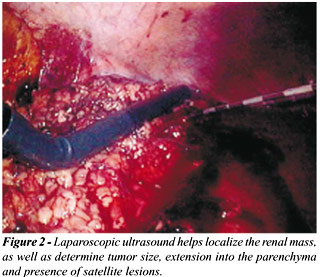
to keep a layer of fascia over the mass (Figure-1). Laparoscopic ultrasonography
can also aid in identifying the location of the renal mass. Detailed information
about tumor size, tumor depth, extension into the parenchyma, distance
from the adjacent calyx, and presence of satellite lesions can be determined
from real time ultrasonography (Figure-2).
The radiofrequency probe, (RITA Medical systems, Mountain View, CA) is
then percutaneously positioned within the mass, and deployed to coagulate
a spherical area including both the lesion and a margin of normal parenchyma
(margin). Settings for the radiofrequency probe for a 3cm lesion are temperature
based, with a target temperature of 105 degrees Celsius, 90 watts, treatment
time of 5.5 minutes, dual cycle. The energy is delivered at 90 W until
the average of the 5 temperature gauges was greater than 105 degrees Celsius,
and then autoregulated to maintain the temperature at this level for 5.5
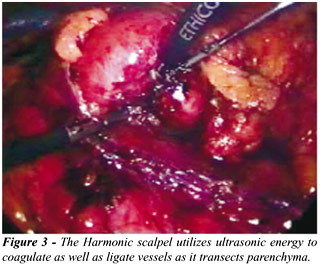
minutes (per the manufacturer’s recommendations). At the conclusion
of the second cycle, the Harmonic scalpel (Ethicon, Cincinnati, OH) is
used to excise the mass together with a 0.5cm margin of normal parenchyma
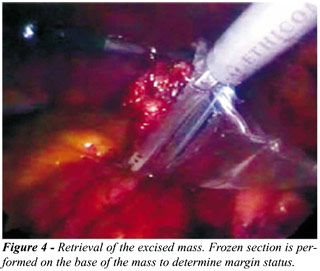
(Figure-3). The lesion is placed in an Endocatch bag, and the parenchymal
resection margin is biopsied (Figure-4). Argon beam coagulation is applied
to the cut the surface. The argon beam is essential for hemostatic control.
Argon is an inert gas that does not support combustion, and is rapidly
cleared from the body. Retrograde injection of indigo carmine dye is performed
to determine collecting system viability. If the collecting system has
been entered, a CT-1 needle with 3-0 polyglactin is used to perform a
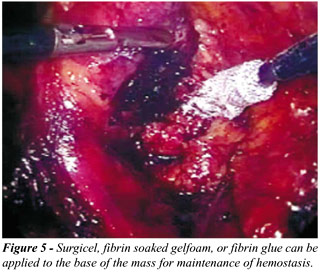
running suture repair. Oxidized cellulose or fibrin glue can be placed
over the resected base to help maintain hemostasis (Figure-5). Follow-up
monitoring includes physical exam, serum creatinine, chest X-ray and abdominal
CT scan at 6 months, and annually thereafter.
DISCUSSION
Laparoscopic
partial nephrectomy was first reported in 1993 by Winfield et al. (3),
in a patient with a lower pole calyceal diverticulum containing a calculus.
Hemostasis was aided through use of a renal tourniquet cinched down around
the lower pole of the kidney. Further application of this concept of parenchymal
compression was investigated by Cadeddu et al. (4), with application of
cable ties circumferentially to the kidney to aid in hemostasis. Reversible,
regional hypoperfusion was achieved in the porcine model. However, in
clinical evaluation of these modalities, adequate hemostasis has been
unreliable with intermittent arterial bleeding from the cut edge of the
kidney despite application of the tourniquet. If excessive force is applied,
as the tourniquet is tightened, cutting, and subsequent fracture of the
renal parenchyma occurs. Alternatively, if the tourniquet is too loose,
significant hemorrhage can occur.
Laparoscopic partial nephrectomy continues
to evolve along 2 therapeutic technical avenues: hilar clamping with ischemia
vs. no hilar clamping. Development of a laparoscopic Satinsky clamp has
achieved en bloc control of the renal hilum in order to allow cold knife
excision of the mass, with laparoscopic repair of the collecting system
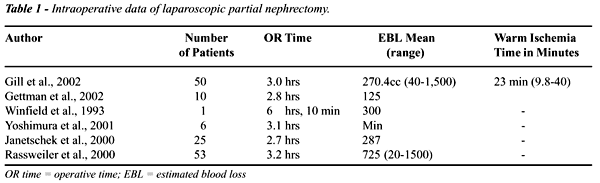
if needed. Gill et al. (5) reported their experience with this technique
in 50 patients, mean tumor size 3.0cm, with warm ischemia time of 23 +
7.4 minutes (range 9.8-40 minutes). Caliceal entry was demonstrated in
18 patients, with immediate repair of the collecting system performed.
Two patients required post operative transfusion, with a mean hospitalization
stay of 2.2 days. Three complications were reported: intraoperative hemorrhage
(n=1), delayed hemorrhage plus nephrectomy (n=1), urine leak (n=1). This
technique is appealing with its goal of duplicating the open surgical
technique. Renal function following this procedure was preserved, with
100% negative margins.
Combination of laparoscopic partial nephrectomy
with ablative techniques has achieved successful excision of renal masses
with adequate hemostasis without hilar clamping. In patients undergoing
excision without hilar control, combination radiofrequency ablation with
immediate excision of the mass has been reported in 10 patients. Mean
tumor size was 2.1cm (range 1.0-3.2cm), mean operative time was 170 minutes
and median blood loss was 125cc. No perioperative complications were reported,
and a final diagnosis of renal cell carcinoma (n=9) and angiomyolipoma
(n=1), with 100% negative margins, was reported. The benefit of hemostasis
without hilar clamping decreases the risk of warm renal ischemia. Furthermore,
excisional partial nephrectomy provides clear pathological analysis and
confirmation of clear margins, and a better oncological approach over
ablative techniques such as cryosurgery or radiofrequency ablation alone.Other
techniques without hilar control have been investigated. Yoshimura et
al. (6) reported use of a microwave tissue coagulator for laparoscopic
partial nephrectomy without hilar clamping. In 6 patients with mean tumor
size of 1.7cm, mean operating time was 186 minutes, blood loss was minimal.
In this approach, multiple insertions of the probe, range 5-23 coagulations,
5-8mm apart were conducted, prior to excision of the mass (Tables 1 and
2).
The benefits of laparoscopy for the kidney
have clearly been demonstrated in terms of less pain, decreased convalescence,
and decreased narcotic requirements. The benchmarks for long term success
of both laparoscopic approaches for radical nephrectomy and partial nephrectomy
will be defined by oncologic principles. Five year outcome data on actuarial
disease free survival will assess the success of these procedures. Janetschek
et al. (7) reported 13.3 month follow-up for laparoscopic radical nephrectomy
and 22.2 month follow-up for wedge resection. One patient had distant
metastases to the lung, a different patient demonstrated multilocular
tumor 1 year postoperatively. There were no local recurrences reported.
For laparoscopic radical nephrectomy, a multi-institutional study of 157
patients reported an actuarial 5 year cancer free rate of 89% for clinical
T2 and 100% for clinical T1 disease (8).
Chan et al. (1) recently reported a comparison
of laparoscopic nephrectomy for renal cell carcinoma to open nephrectomy.
At follow-up of 35.6 months vs. 44 months, respectively, no statistical
difference was determined on Kaplan Meier actuarial survival analysis.
Patients were matched for age and side, with mean tumor size 5.1cm (1-13cm).
Clearly, the laparoscopic radical nephrectomy for T1/T2 lesions is equivalent
to that of open surgery in both efficiency and efficacy.
Laparoscopic radical nephrectomy for renal
cell carcinoma has clearly demonstrated low morbidity and equivalent cancer
control. The rates for local recurrences and metastatic spread are low
and actuarial survival high. Furthermore, laparoscopic partial nephrectomy
has demonstrated to be technically feasible, with low morbidity. With
short term outcomes demonstrating laparoscopic partial nephrectomy as
an efficacious procedure, the role of laparoscopic partial nephrectomy
should continue to increase.
REFERENCES
- Chan DY, Cadeddu JA, Jarrett TW, Marshall FF, Kavoussi LR: Laparoscopic radical nephrectomy: cancer control for renal cell carcinoma. J Urol. 2001; 166: 2095-100.
- Montie JE, Novick AC: Partial nephrectomy for renal cell carcinoma. J Urol. 1992; 148:1835.
- Winfield HN, Donovan JF, Godet AS, Clayman RV: Laparoscopic partial nephrectomy: Initial case report for benign disease. J Endourol. 1993; 7:521-6.
- Cadeddu JA, Corwin TS, Traxer O, Collick C, Saboorian HH, Pearle MS: Hemostatic laparoscopic partial nephrectomy: cable-tie compression. Urology 2001; 5:562-6.
- Gill IS, Desai MM, Kaouk JH, Meraney AM, Murphy DP, Sung GT, et al.: Laparoscopic partial nephrectomy for renal tumor: duplicating open surgical techniques. J Urol. 2002; 167:469-76.
- Yoshimura K, Okubo K, Ichioka K, Terada N, Matsuta Y, Arai Y: Laparoscopic partial nephrectomy with a microwave tissue coagulator for small renal tumor. J Urol. 2001; 165:1893-6.
- Janetschek G, Jeschke K, Peschel R: Laparoscopic surgery for stage 1 renal cell carcinoma: Radical nephrectomy and wedge resection. Eur Urol. 2000; 38:131.
- Cadeddu JA, Ono Y, Clayman RV, Barrett PH, Janetschek G, McDougall EM, et al.: Laparoscopic nephrectomy for renal cell carcinoma: Evaluation of efficacy and safety. A multi center experience. Urology 1998; 52:773-7.
- Gettman MT, Bishoff JT, Su LM, Chan D, Kavoussi LR, Jarrett TW, et al.: Hemostatic laparoscopic partial nephrectomy: initial experience with the radiofrequency coagulation-assisted technique. Urology 2001; 58:8-11.
- Rassweiler JJ, Abbou C, Janetschek G, Jeschke K: Laparoscopic partial nephrectomy. The European experience. Urol Clin North Am. 2000; 27:721-36.
____________________
Received: May 20, 2002
Accepted: April 20, 2002
_______________________
Correspondence address:
Dr. Benjamin R. Lee
Department of Urology
Long Island Jewish Medical Center
New Hyde Park, New York, 11040, USA
Fax: + 1 718 343-6254
E-mail: blee@lij.edu