RESISTANCE
OF HUMAN SPERMATOZOA TO CRYOINJURY IN REPEATED CYCLES OF THAW-REFREEZING
( Download pdf )
SIDNEY VERZA JR., CINTHIA M. FEIJO, SANDRO C. ESTEVES
ANDROFERT, Referral Center for Male Reproduction, Campinas, Sao Paulo, Brazil
ABSTRACT
Objective:
To study the resistance of human spermatozoa to cryoinjury in repeated
cycles of thaw-refreezing by using the fast liquid nitrogen vapor method.
Materials and Methods: Semen specimens were obtained from sixteen normal
and oligozoospermic individuals who required disposal at the sperm bank.
Five of them had testicular cancer. Specimens were thawed and an aliquot
was removed for analysis. The remaining specimens were refrozen without
removing the cryomedia. Repeated freeze-thaw cycles were performed until
no motile sperm were observed. Sperm motility, number of motile spermatozoa
and viability were determined after thawing. Resistance to cryoinjury
was compared between groups and also after each refreezing cycle within
groups.
Results: Motile spermatozoa were recovered after five and two refreeze-thawing
cycles in normozoospermic and oligozoospermic specimens, respectively.
There were no significant differences in the recovery of motile spermatozoa
between thaws within each group of normal and oligozoospermic specimens,
but percentage motility and total number of motile spermatozoa were significantly
lower in the oligozoospermic one. Specimens from men with cancer were
exposed to six refreeze-thawing cycles. Although recovery of motile spermatozoa
was significantly impaired after each thawing, there were no significant
differences in the recovery of motile sperm between thaws in cancer and
non-cancer groups.
Conclusions: Human spermatozoa resist repeated cryopreservation using
the fast liquid nitrogen vapor method. Normozoospermic specimens withstand
refreezing for an average two cycles longer than oligozoospermic ones.
Specimens from cancer patients seem to resist repeated cryoinjury similarly
to non-cancer counterparts. Resistance to repeated cryoinjury was related
to the initial semen quality.
Key
words: sperm; fertility; cryopreservation; oligozoospermia; cancer
Int Braz J Urol. 2009; 35: 581-91
INTRODUCTION
Since the
1960´s, sperm banking has been successfully used to preserve male
fertility (1). Efforts have been made to improve cryopreservation of human
spermatozoa and to obtain better gametes after thawing. It is well known
that the freeze-thawing process affects the fertile potential of human
sperm on several aspects. The freeze and thawing process decrease sperm
motility (2) and sperm penetration into the cervical mucus (3), alter
cell membrane fluidity (4,5), decrease acrosome integrity (6) and may
induce sperm apoptotic deoxyribonucleic acid (DNA) fragmentation (7).
Lower fertilization and pregnancy rates are achieved when frozen-thawed
spermatozoa are used for intrauterine insemination (8) and conventional
in-vitro fertilization (9). However, it has been shown that similar fertilization
and pregnancy rates can be obtained with intracytoplasmic sperm injection
(ICSI) using both frozen-thawed and fresh motile spermatozoa (10).
Indications for sperm banking have expanded in the recent years, but the
group of cancer patients in reproductive age deserves special attention.
Chemotherapy and radiation are often gonadotoxic and although fertility
restoration occurs in about 50% of cancer survivors, permanent sterility
is common (11,12). Therefore, sperm cryopreservation is recommended before
therapy (13,14). The main concern, however, is that cancer patients are
often urged to start treatment and in most individuals only very few specimens
preserved.
Even using modern assisted reproductive techniques (ART), pregnancy rates
per attempt are below the 50% rate (10,15,16). Then, many couples require
multiple ART attempts in order to achieve a live birth. When frozen-thawed
sperm are used for ART, one or more vials have to be thawed until an adequate
number of viable sperm are obtained. However, ART procedures require very
few gametes, especially ICSI, and the remaining ones are left over and
ultimately discharged. Refreezing thawed specimens would provide additional
opportunities for conception in men who have banked a limited number of
sperm specimens, such as cancer patients and those who have a small supply
of donor semen left from a previous pregnancy and wish to use it for siblings.
The objective of this study was to assess the resistance of human spermatozoa
to cryoinjury in repeated cycles of thaw-refreezing.
MATERIALS AND METHODS
Specimens
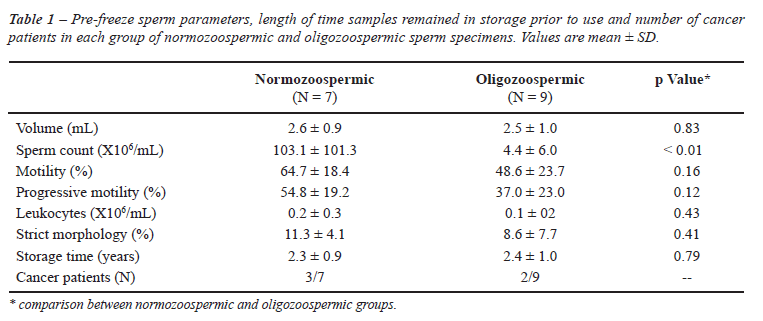
Frozen semen specimens were obtained from 16 subjects who requested disposal from our sperm bank. We included all eligible patients who requested disposal over a 6-year period. Specimens remained in storage for an average time of 2.3 years (interval: 1-6 years). According to the pre-freeze sperm count at the time of banking, specimens were divided as normozoospermic (= 20X106/mL; n = 7) or oligozoospermic (< 20X106/mL; n = 9). Reasons for sperm banking were: (i) enrollment in our ART egg donation program, in which sperm cryopreservation is mandatory to facilitate patient treatment synchronization (n = 8); (ii) vasectomy (n = 3), and cancer (n = 5). All cancer patients had testicular malignancies. Two of them have previously had unilateral orchiectomy at the time of banking and were oligozoospermic, while three banked sperm before surgery and were normozoospermic. We have also divided the specimens according to the existence of cancer (n = 5) or not (n = 11), in order to compare the resistance of repeated freezing and thawing between them. The study was approved by our Institutional Review Board and informed consent was obtained from all subjects.
Cryopreservation and
Thawing Procedures
At the time of initial freezing, specimens were collected by masturbation after 2-3 days of ejaculatory abstinence into sterile containers. An aliquot of each liquefied specimen was analyzed according to the WHO guidelines (17). Specimens were cryopreserved by the liquid nitrogen vapor method, using TEST-yolk buffer with glycerol as a freezing media, as previously described (4). In brief, a vial of the freezing agent (TEST-yolk buffer with 12% glycerol, Irvine Scientific, USA) was thawed by incubation at 37°C. An aliquot of the medium equal to 25% of the original specimen volume was then added to the specimen. The specimen was gently mixed for 5 minutes using an aliquot mixer. This process was repeated until the ratio of freezing medium to ejaculate was 1:1 v/v. The mixture was loaded into 1.0-mL cryovials, which were placed at -20°C for 8 minutes and submerged in liquid nitrogen vapor at -79°C for 2 hours. The vials were then plunged into liquid nitrogen for long-term storage at -196°C. For thawing, all cryovials from each subject were removed from liquid nitrogen storage dewars and were thawed at room temperature for 5 minutes. Then, cryovials were transferred to a 37°C incubator for 20 minutes (4).
Assessment of Sperm
Parameters
After incubation and homogenization of frozen-thawed specimens of each individual patient, an aliquot was removed for analysis of sperm motility and total number of motile sperm. Recovery percentages of motile spermatozoa were calculated after each thaw as the ratio between post-thaw and pre-freeze percentages of motile sperm X100. If only immotile sperm were seen after thawing, testing for sperm viability was performed using eosin exclusion dye (17).
Refreeze Procedure
After removing an aliquot for analysis, the remaining specimen was refrozen by the same freezing method described above, neither removing the cryoprotectant used in the original freezing cycle nor adding new one. The specimens were then stored in liquid nitrogen for at least 48 hours and thawed using the method previously described. The thaw-refreeze cycles were repeated until no motile sperm were obtained.
Statistical Analysis
Data are expressed as mean ± SD. The Student’s unpaired t-test was used to analyze statistical differences in pre-freeze sperm parameters and in percentage motility, percentage recovery of motile sperm and total number of motile spermatozoa in normozoospermic and oligozoospermic groups, and also in cancer and non-cancer specimens after each thaw. The Student’s paired t-test was used to compare percentage motility, percentage recovery of motile sperm and total number of motile spermatozoa before and after each refreezing cycle within groups. Data were tested for homogeneity and normality by the Kolmogorov-Smirnov test. An alpha level of 0.05 or less was considered significant. Statistical analysis was performed using the Statistica® software package (USA).
RESULTS
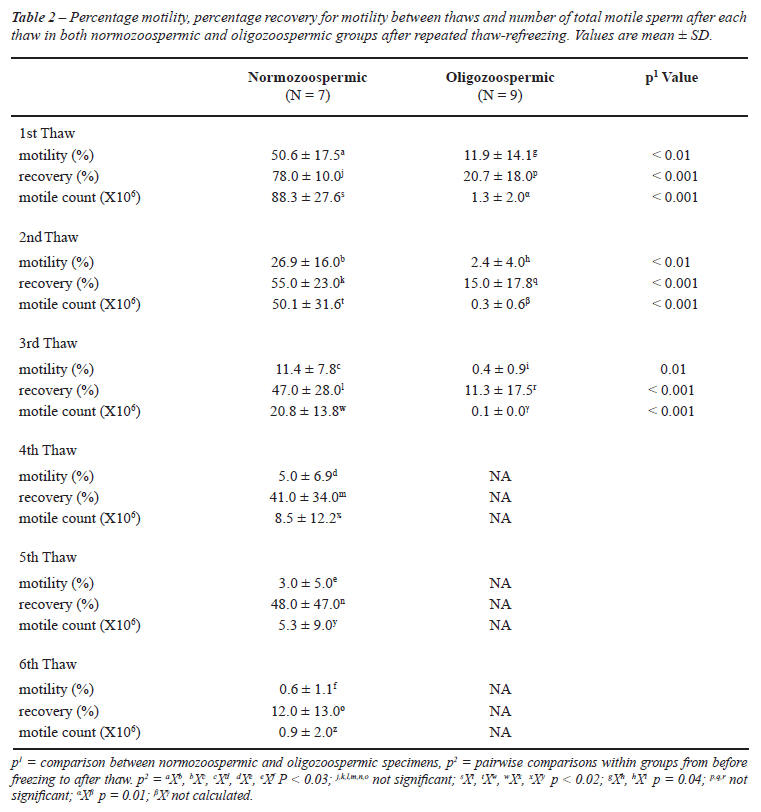
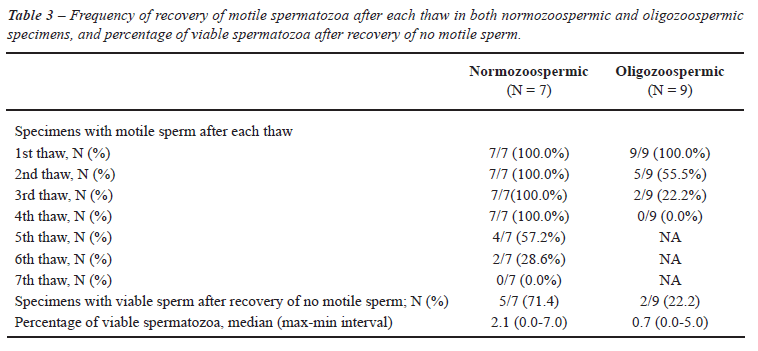
The mean ± SD age of normozoospermic and oligozoospermic individuals were 36.0 ± 8.0 and 29.9 ± 4.2 years, respectively (P = 0.52). The length of time samples remained in storage prior to use was not different between groups (Table-1). Pre-freeze sperm parameters are shown in Table-1. Recovery of motile spermatozoa was observed through 5 and 2 refreeze-thawing cycles in normozoospermic and oligozoospermic specimens, respectively (Table-2). A significant decrease in sperm motility and number of total motile spermatozoa was observed after each refreeze-thawing cycle in both normozoospermic and oligozoospermic groups (Table-2, p < 0.05). There were no significant differences in percentage recovery of motile sperm between thaws within each group of normozoospermic and oligozoospermic specimens (Table-2). However, percentage motility, percentage recovery of motile sperm and total number of motile spermatozoa were significantly lower in the oligozoospermic group when compared to the normozoospermic one (Table-2, p < 0.001). Motile spermatozoa were found in all normozoospermic specimens up to the third refreeze-thawing cycle. Approximately 50% and 25% of all normozoospermic specimens had motile sperm after the 4th and 5th refreeze-thawing, respectively. However, only 55% of the specimens had motile spermatozoa after the first refreeze-thawing cycle in the oligozoospermic group (Table-3). Viable spermatozoa were still detected in 71.4% (5/7) normozoospermic and 22.2% (2/9) oligozoospermic specimens when only immotile gametes were found after thawing (Table-3).
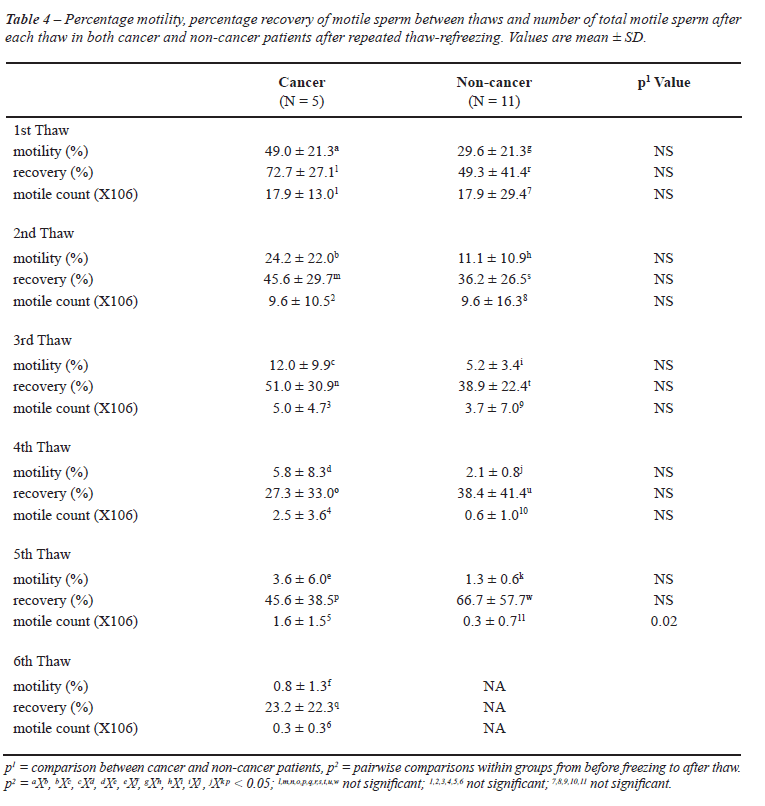
The mean ± SD age of cancer and non-cancer patients were 26.4 ±
6.0 and 32.7 ± 8.3 years, respectively (p = 0.03). Pre-freeze sperm
parameters of cancer and non-cancer specimens were not statistically different,
except for sperm motility that was higher in the cancer group (62.5 ±
18.4 versus 51.9 ± 21.4, p = 0.04). A significant decrease in sperm
motility and number of total motile spermatozoa was seen after each refreeze-thawing
cycle in both groups, but there were no significant differences in percentage
recovery of motile sperm between thaws within each group of cancer and
non-cancer specimens (Table-4). Moreover, percentage motility, motility
recovery and total number of motile spermatozoa were comparable in cancer
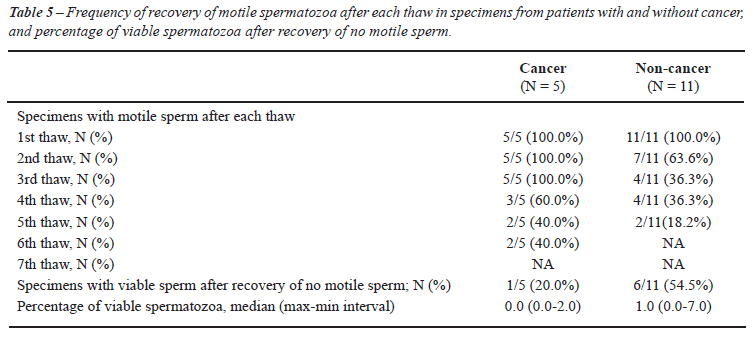
and non-cancer groups (Table-4). Recovery of motile spermatozoa was seen
through six and five refreeze-thawing cycles in specimens from individuals
with and without cancer, respectively (Tables 4 and 5).
COMMENTS
Cryosurvival
of human sperm depends mainly on the cryoprotectant, the freezing technique
and the initial quality of the specimen. The use of glycerol to prevent
injury to human spermatozoa during cryopreservation is well established
(18), and its association with buffers, such as Tris (hydroximethyl amino
metano) and TES (n-Tris [hydroximethyl] methyl-2-amino-ethane sulphonic
acid), and egg-yolk yield optimal cryosurvival rates (19). Slow freezing
using programmable freezing machines or fast freezing, as used in this
study, seems to have no direct effect on thaw survival both in normal
and poor quality sperm (20). It has been demonstrated that, independently
from the freezing technique, motility from poor quality sperm is kept
constant during the first 3 hours after thawing, but it is drastically
reduced by the end of an incubation period of 24 hours (20). In this study,
we used a rapid vapor freezing method because it is less expensive, time-consuming
and labor-intensive, and it has proven equally effective in the recovery
of post-thaw motile sperm (20,21).
Studies focusing on the resistance of human spermatozoa to cryoinjury
after repeated thaw-refreezing cycles are scarce. Polcz et al., studying
only normozoospermic men, first demonstrated the ability of human spermatozoa
to resist cryoinjury in successive thaw-refreeze cycles (22). They observed
that spermatozoa were able to withstand five thaw-refreeze cycles and
still maintain motility and vitality, although a marked reduction in motility
occurred. After the 3rd, 4th and 5th thaw-refreeze cycle, only 3.5%, 1.5%
and 1.8% motile sperm were seen, respectively. In another study, it has
been shown that spermatozoa could resist up to seven cycles thaw-refreeze
(23), although a linear decrease of motility per cycle was observed. These
authors also compared slow and fast freezing techniques, and concluded
that fast freezing preserved motility for an average of 2.75 cycles more
than slow refreezing. However, their study differs from ours because no
information was provided regarding the quality of sperm specimens and
slow controlled-freezing was used for the initial freezing cycle instead
of the fast liquid nitrogen vapor method. Bandularatne & Bongso (21)
evaluated the extent of sperm cryoinjury up to three repeated freezing
in normozoospermic and oligozoospermic men using both slow and fast freezing
techniques. They observed a significant reduction in the recovery of motile
and viable sperm after each thaw independent of the freezing method. Differences
in the recovery of motile and viable sperm between oligozoospermic and
normozoospermic specimens were not observed (21).
We also evaluated the resistance to cryoinjury in normozoospermic and
oligozoospermic sperm samples, as did Bandularatne & Bongso (21),
but we performed repeated freeze-thawing until absence of post-thaw motile
sperm was seen. We observed that recovery of motile spermatozoa was significantly
impaired after each refreeze-thawing cycle, but motile sperm was still
found after five and two refreezing cycles in normozoospermic and oligozoospermic
groups, respectively. Therefore, sperm from normozoospermic men withstood
repeated freezing longer than oligoozoospermic ones. Also, there were
significant differences in the recovery of motile sperm between thaws
when normozoospermic and oligozoospermic groups were compared. We found
that the percentage of motile spermatozoa dropped nearly to half after
each subsequent freeze-thawing process in normozoospermic specimens, but
dropped considerably greater in oligozoospermic ones. A possible explanation
for the different results between Bandularatne and Bongso’s study
and ours may be the fact that they used higher glycerol concentration.
In a previous report, we have shown that sperm cryosurvival may be optimized
when cryomedia containing 15% glycerol was used when compared to the standard
12% (21). In addition, there was a marked difference in the degree of
oligozoospermia between studies. Our oligozoospermic group had very low
sperm counts (mean count 4.4 million/mL) while theirs had only mild oligozoospermia
(mean 15.7 million/mL). Moreover, mean pre-freeze sperm motility was 48.6%
in our group when compared to 73.4% in theirs. Given that sperm cryosurvival
is partially dependent on the semen quality before freezing (13,25,26),
we speculate that sperm produced by men with severely defective spermatogenesis
is more sensitive to cryoinjury than their normal counterpart is.
Pre-freeze motility of = 15% can predict a post-thaw motility of >
10% with > 75% accuracy (27). Based on these findings, along with the
effectiveness of ICSI to achieve successful fertilization with very few
sperm, cryopreservation is recommended even when a limited number of motile
sperm is available (27). In our study, post-thaw recovery of motile sperm
that would be adequate for ICSI was obtained even with pre-freeze motility
< 12%. Interestingly, all normozoospermic specimens with pre-freeze
motility > 10% had motile sperm after thawing, and pre-freeze motility
of 5% and 3% yielded recovery of motile sperm after thawing in 50% and
25% of the specimens, respectively. Using the same cutoff point of >
10%, only 50% of oligozoospermic specimens had motile sperm after thawing.
From a clinical standpoint, it would be ideal to compare the results of
normal and abnormal samples from men with and without cancer to determine
whether cancer patients do indeed fare worse than normozoospermic/oligozoospermic
non-cancer patients do. Although our sample size did not allow such comparisons,
we have grouped all cancer and non-cancer patients and compared the resistance
to repeat freeze-thawing. We found that semen specimens from men with
cancer withstood repeated cryoinjury similarly to their counterparts without
cancer. Although the recovery of motile spermatozoa was significantly
impaired after each refreeze-thawing cycle, there were no significant
differences in the recovery of motile sperm between thaws when cancer
and non-cancer groups were compared. From our data, it seems that the
resistance for repeated cryoinjury is related to the initial semen quality
before cryopreservation. However, these results should be interpreted
with caution because our cancer population consisted of only five men,
and three of them had very good quality semen at the time of banking.
Although it is clear from this study and others that human sperm can resist
repeated freezing, the fertilizing ability of such gametes to produce
a viable and healthy offspring has not yet been properly addressed. To
date, function of refrozen and thawed human spermatozoa has been assessed
only by microinjection into zona-intact hamster oocytes (Hamster Intracytoplasmic
Sperm Injection test [HICSI test]) (21). Similar fertilization rates were
obtained up to the third refreezing cycle when refrozen and fresh spermatozoa
were compared (22.2% versus 27.3%, respectively) (21). HICSI assay seems
to be a reliable indicator for fertilization, but it is not appropriate
to predict the occurrence of a viable pregnancy (28). It has been demonstrated
that the cryopreservation process induces DNA damage, and that DNA fragmentation
is higher in poor quality specimens, such as from oligozoospermic men,
when compared to normal ones (7). Oocyte ability to repair double-strand
sperm DNA fragmentation is limited, and this type of fragmentation may
lead to genomic mutation with consequent embryo alteration. A recent study
has shown a significant increase in the percentage of spermatozoa exhibiting
DNA fragmentation following the first cycle of refreezing and thawing
(29). This study examined the resistance of human sperm up to three refreezing
cycles using a slow controlled rate method, and concluded that up to three
refreezing cycles can be performed with a level of risk to sperm DNA comparable
to that following a single cycle of freezing and thawing, provided that
samples are refrozen in their original cryoprotectant and not washed.
Therefore, great caution is needed if ICSI is to be performed with thaw-refrozen
sperm, and the safety of this procedure has to be extensively studied.
In our study, specimens were cryopreserved with the seminal plasma, and
the cryoprotectant used in the first freezing cycle remained throughout
the experiment. We observed that refreezing human sperm without processing
and/or removing or adding cryoprotectant was fast, simple, and inexpensive.
Although we have not examined the effects of removing the seminal plasma
before freezing or after thawing on motility, or the role of removing
the cryoprotectant and adding a new one after each thawing cycle, Polcz
et al. (22) reported marked declines in motility and viability when these
washing steps were included and fresh cryoprotectant media was added.
Moreover, Thomson et al. (2009) reported that the level of DNA fragmentation
more than doubled when the samples were washed and fresh cryoprotectant
was added after each thaw, but it only increased slightly when samples
were refrozen in the original cryoprotectant without any further treatment
(29). In another study by the same authors, a comparable increase in the
percentage sperm DNA fragmentation post-cryopreservation was observed
both with and without the addition of cryoprotectant (30). Their results
seem to indicate that the cryoprotectant plays no role in the generation
of DNA damage during cryopreservation. On the other hand, the washing
steps, which involve dilution and centrifugation, subject the already
compromised thawed spermatozoa to many cycles of osmotic shock that may
cause mechanical damage to cellular structures and possibly to the DNA
molecule (27,29,30). The observed increase in the level of sperm DNA damage
and decrease in motility and viability after the freeze-thawing process
may be due to a combination of factors, such as the oxidative stress generated
via lipid peroxidation, the depletion of protective seminal and spermatozoa
antioxidants or the process of cryopreservation itself (27). Removal of
seminal fluid via washing steps following the freeze-thawing process may
thus deplete protective seminal antioxidants and increase the susceptibility
of spermatozoa to oxidative stress.
CONCLUSION
Human spermatozoa can resist cryoinjury after repeated cycles of cryopreservation using the fast vapor freezing method. Normozoospermic specimens withstand refreezing for an average 2 cycles longer than oligozoospermic ones. Specimens from cancer patients seem to resist repeated cryoinjury similarly to non-cancer counterparts. Sperm ability to resist injury due to the thawing-refreeze process appears to be related to the initial semen quality. Due to the low number of motile sperm and reduced post-thaw motility, gametes that survived repeated freezing would be suitable for intracytoplasmic sperm injection only. Refreezing leftover frozen-thawed specimens may be recommended for patients who were not able to freeze multiple specimens, such as cancer patients and those individuals who have limited supply of donor semen left from a previous pregnancy and wish to use it for siblings. Further studies are required to assess the fertility potential and safety of the use of refrozen human semen for assisted reproduction.
Acknowledgement
Mrs. Fabiola Bento edited the manuscript.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Sherman JK: Low temperature research on spermatozoa and eggs. Cryobiology. 1964; 1: 103-29.
- Centola GM, Raubertas RF, Mattox JH: Cryopreservation of human semen. Comparison of cryopreservatives, sources of variability, and prediction of post-thaw survival. J Androl. 1992; 13: 283-8.
- Fjällbrant B, Ackerman DR: Cervical mucus penetration in vitro by fresh and frozen-preserved human semen specimens. J Reprod Fertil. 1969; 20: 515-7.
- Esteves SC, Sharma RK, Thomas AJ Jr, Agarwal A: Suitability of the hypo-osmotic swelling test for assessing the viability of cryopreserved sperm. Fertil Steril. 1996; 66: 798-804.
- Esteves SC, Sharma RK, Thomas AJ Jr, Agarwal A. Evaluation of acrosomal status and sperm viability in fresh and cryopreserved specimens by the use of fluorescent peanut agglutinin lectin in conjunction with hypo-osmotic swelling test. Int Braz J Urol. 2007; 33: 364-74.
- Cross NL, Hanks SE: Effects of cryopreservation on human sperm acrosomes. Hum Reprod. 1991; 6: 1279-83.
- de Paula TS, Bertolla RP, Spaine DM, Cunha MA, Schor N, Cedenho AP: Effect of cryopreservation on sperm apoptotic deoxyribonucleic acid fragmentation in patients with oligozoospermia. Fertil Steril. 2006; 86: 597-600.
- Peterson EP, Alexander NJ, Moghissi KS: A.I.D. and AIDS--too close for comfort. Fertil Steril. 1988; 49: 209-10.
- Szczygiel MA, Kusakabe H, Yanagimachi R, Whittingham DG: Intracytoplasmic sperm injection is more efficient than in vitro fertilization for generating mouse embryos from cryopreserved spermatozoa. Biol Reprod. 2002; 67: 1278-84.
- Kuczynski W, Dhont M, Grygoruk C, Grochowski D, Wolczynski S, Szamatowicz M: The outcome of intracytoplasmic injection of fresh and cryopreserved ejaculated spermatozoa--a prospective randomized study. Hum Reprod. 2001; 16: 2109-13.
- Lass A, Akagbosu F, Abusheikha N, Hassouneh M, Blayney M, Avery S, et al.: A programme of semen cryopreservation for patients with malignant disease in a tertiary infertility centre: lessons from 8 years’ experience. Hum Reprod. 1998; 13: 3256-61.
- Naysmith TE, Blake DA, Harvey VJ, Johnson NP: Do men undergoing sterilizing cancer treatments have a fertile future? Hum Reprod. 1998; 13: 3250-5.
- Hallak J, Kolettis PN, Sekhon VS, Thomas AJ Jr, Agarwal A: Cryopreservation of sperm from patients with leukemia: is it worth the effort? Cancer. 1999; 85: 1973-8.
- Agarwal A, Ranganathan P, Kattal N, Pasqualotto F, Hallak J, Khayal S, et al.: Fertility after cancer: a prospective review of assisted reproductive outcome with banked semen specimens. Fertil Steril. 2004; 81: 342-8.
- Esteves SC, Schneider DT, Verza S Jr. Influence of antisperm antibodies in the semen on intracytoplasmic sperm injection outcome. Int Braz J Urol. 2007; 33: 795-802.
- Verza Jr S, Esteves SC: Sperm defect severity rather than sperm source is associated with lower fertilization rates after intracytoplasmic sperm injection. Int Braz J Urol. 2008; 34: 49-56.
- World Health Organization (WHO): Laboratory Manual for the examination of human semen and sperm-cervical mucus interaction. 4th ed., Cambridge, Cambridge University Press. 1999; pp. 68-70.
- Hammitt DG, Walker DL, Williamson RA: Concentration of glycerol required for optimal survival and in vitro fertilizing capacity of frozen sperm is dependent on cryopreservation medium. Fertil Steril. 1988; 49: 680-7.
- Hallak J, Sharma RK, Wellstead C, Agarwal A: Cryopreservation of human spermatozoa: comparison of TEST-yolk buffer and glycerol. Int J Fertil Womens Med. 2000; 45: 38-42.
- Esteves SC, Spaine DM, Cedenho AP, Srougi M: Effects of the technique of cryopreservation and dilution/centrifugation after thawing on the motility and vitality of spermatozoa from oligozoospermic men Int Braz J Urol. 2003; 29: 133-140.
- Bandularatne E, Bongso A: Evaluation of human sperm function after repeated freezing and thawing. J Androl. 2002; 23: 242-9.
- Polcz TE, Stronk J, Xiong C, Jones EE, Olive DL, Huszar G: Optimal utilization of cryopreserved human semen for assisted reproduction: recovery and maintenance of sperm motility and viability. J Assist Reprod Genet. 1998; 15: 504-12.
- Rofeim O, Brown TA, Gilbert BR: Effects of serial thaw-refreeze cycles on human sperm motility and viability. Fertil Steril. 2001; 75: 1242-3.
- Verza Jr. S, Feijo CM, Schneider DT, Esteves SC: Human cryopreservation: Comparison of three cryoprotectant solutions. Fertil Magazine. 2007; 7: 26-8.
- Esteves SC, Spaine DM, Cedenho AP: Effects of pentoxifylline treatment before freezing on motility, viability and acrosome status of poor quality human spermatozoa cryopreserved by the liquid nitrogen vapor method. Braz J Med Biol Res. 2007; 40: 985-92.
- Donnelly ET, McClure N, Lewis SE: Cryopreservation of human semen and prepared sperm: effects on motility parameters and DNA integrity. Fertil Steril. 2001; 76: 892-900.
- Padron OF, Sharma RK, Thomas AJ Jr, Agarwal A: Effects of cancer on spermatozoa quality after cryopreservation: a 12-year experience. Fertil Steril. 1997; 67: 326-31.
- Gvakharia MO, Lipshultz LI, Lamb DJ: Human sperm microinjection into hamster oocytes: a new tool for training and evaluation of the technical proficiency of intracytoplasmic sperm injection. Fertil Steril. 2000; 73: 395-401.
- Thomson LK, Fleming SD, Barone K, Zieschang JA, Clark AM: The effect of repeated freezing and thawing on human sperm DNA fragmentation. Fertil Steril. 2009 [Epub ahead of print].
- Thomson LK, Fleming SD, Schulke L, Barone K, Zieschang JA, Clark AM: The DNA integrity of cryopreserved spermatozoa separated for use in assisted reproductive technology is unaffected by the type of cryoprotectant used but is related to the DNA integrity of the fresh separated preparation. Fertil Steril. 2009; 92:991-1001.
____________________
Accepted after revision:
April 6, 2009
_______________________
Correspondence address:
Dr. Sandro C. Esteves
Av. Dr. Heitor Penteado, 1464
Campinas, SP, 13075-460, Brazil
Fax: + 55 19 3294-6992
E-mail: s.esteves@androfert.com.br
EDITORIAL COMMENT
This is an interesting study with the most significant finding being that spermatozoa from cancer patients is capable of withstanding 6 freeze-thaw cycles using the fast liquid nitrogen vapor method. This is important for those cancer patients who are only able to freeze a small amount of semen prior to undergoing treatment, providing more hope for fathering children in their future. The authors also report that the spermatozoa from oligozoospermic men is capable of withstanding significantly fewer freeze-thaw cycles than normozoospermic men and conclude that repeated freezing and thawing is a feasible practice using their method of freezing and that the resistance to cryoinjury is related to the initial semen quality. The main limitation of this study is the relatively low sample size studied and further studies involving cancer patients are needed to confirm these promising results.
Dr.
Laura Kelly Thomson
Fertility First, Hurstville
Westmead Hospital
University of Sydney
Hurstville, Sydney Australia
E-mail: lthomson@fertilityfirst.com.au